Asthma is a world-recognized medical problem. It is listed as one of the four stubborn diseases by the World Health Organization. The number of people suffering from asthma continues to increase every year in the world.
In order to help more patients in developing countries treat asthma or chronic obstructive pulmonary disease, Indian medical and home health company giant Zydus Cadiila and IDC of the United Kingdom have joined forces to design a drug delivery device for asthma treatment that is more effective, easier to use and lower cost.
The research and development of asthma inhalers is extremely complex and costly. The world's large pharmaceutical companies and medical equipment manufacturers have almost monopolized the patents of such products on the market. How to avoid the existing patents on the market, form their own product characteristics, and also take into account the development The difficulty of cost control is the difficulty of developing and designing this product.
The IDC product design team in the UK has planned a complete and detailed design and development process based on our classic four-stage product design process.
In the early stage of product definition, it is an important link for foreign enterprises to invest in the research and analysis of existing patents in the market in the process of developing and designing such products, because this is the premise of realizing their own technological innovation. Only on this basis, enterprises can discover the patent window, realize the development of new product features, and finally bring commercial returns to the enterprise by owning invention patents.
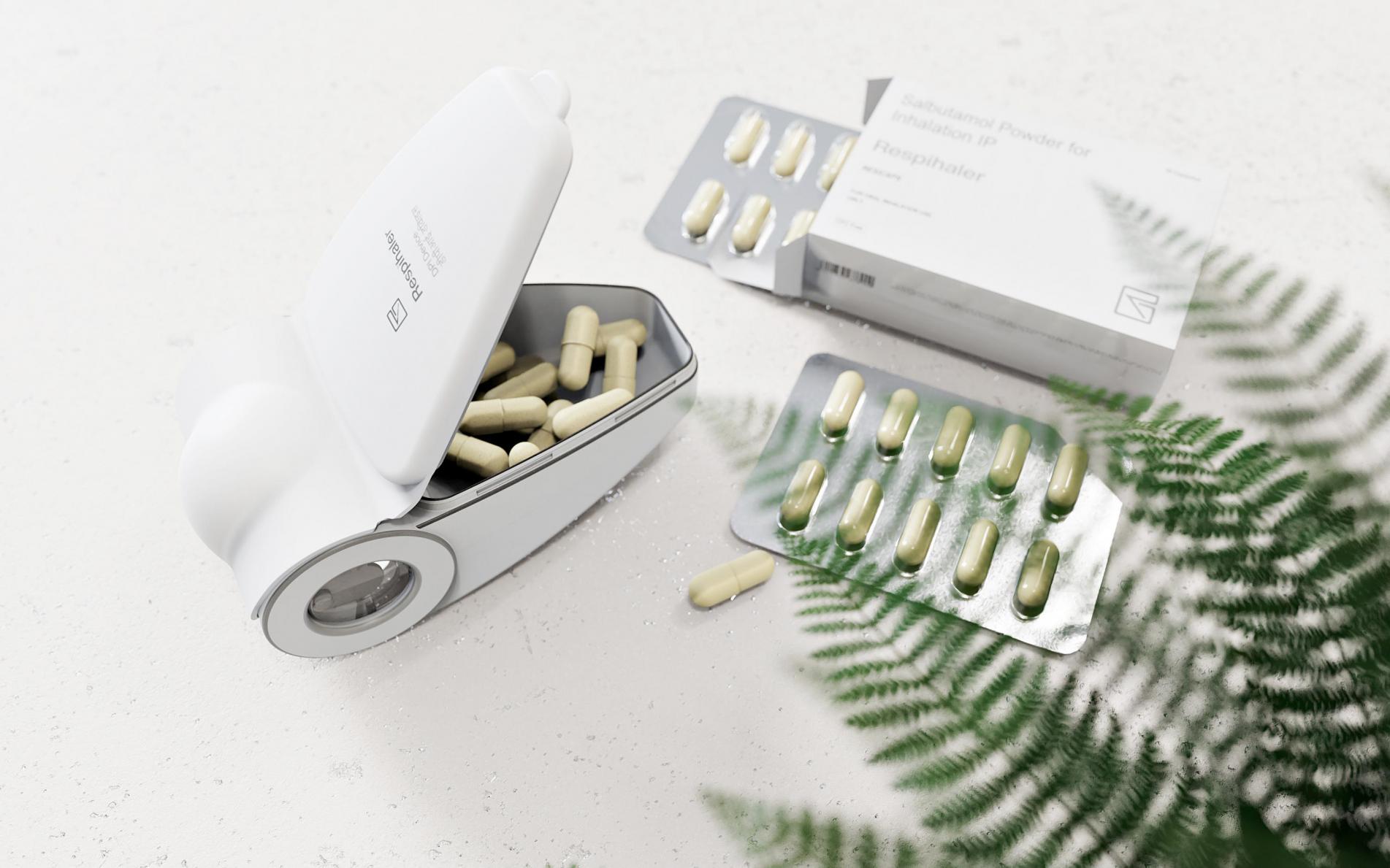
The asthma treatment drug delivery device designed and developed by IDC in the United Kingdom is aimed at capsule administration.
After a large number of patent analysis, the team successfully simplified the user steps and realized a simple and few-step capsule opening (punctured) method in the device, I .e. the capsule was sent into the cavity while the capsule wall was broken, so that the capsule and the powder entered the cavity at the same time for administration. This parallel opening method of pushing in and breaking the capsule not only makes the steps of taking medicine more concise, but also cleverly avoids the restrictions of existing patents in the market and discovers a new invention patent point for the product.
Another important function of such drug delivery devices is the achievement of high drug delivery efficiency.
First, given that asthma patients usually have weak lung function, the airflow pressure they breathe in through the inhaler is relatively small. Secondly, inhalation powder particles should be as small as possible to facilitate absorption. After repeated tests, the final solution designed by IDC in the United Kingdom controls the vortex motion of the airflow in the cavity by ensuring the continuous airflow pressure during inhalation, so that the collision motion between particles and between particles and walls is sufficient, and the particles can be crushed. The particles can fully collide to a finer state before entering the human body. In addition, the more ergonomic design of the medicine mouth greatly improves the drug delivery efficiency of the human body ventilator.
Adhering to the principle of simplicity (or simplification) in the design and development of all products, the IDC product design team in the UK has streamlined the structural parts of the product and realized a complete assembly with only 5 plastic parts. The use of high-quality inhalers, making the product intuitive and easy to use, easy to operate, and also meet the market requirements for low cost.
Case link: https://www.idcdesigncn.com/medical/respihaler/

Product design for third world or developing countries needs to be cost-controlled with superior performance



Fluid analysis and simulation using CFD to facilitate product design and production optimization
本作品版权归 英国IDC产品设计 所有,禁止匿名转载及个人使用,任何商业用途均需联系原作者。

新用户?创建账号
登录 重置密码

请输入电子邮件以重置密码。
Pretty good!
High technology
Practical